Rezpegaldesleukin Shows Promising Efficacy Across Disease Severity in Atopic Dermatitis Phase 2b Trial
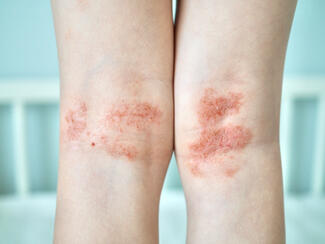
A phase 2b trial of rezpegaldesleukin, a first-in-class regulatory T-cell (Treg)–enhancing biologic, demonstrates significant clinical improvements in moderate-to-severe atopic dermatitis (AD), with consistent efficacy across disease severity levels.
“Rezpegaldesleukin is a novel, first-in-class biologic targeting regulatory T cells (Tregs),” the investigators noted, highlighting its unique immunomodulatory mechanism.
In this randomized, placebo-controlled study, 393 patients were assigned to multiple rezpegaldesleukin dosing regimens or placebo during a 16-week induction period. The primary endpoint was percent change in Eczema Area and Severity Index (EASI) score, with additional outcomes including EASI-75, EASI-90, Validated Investigator Global Assessment (vIGA) response, and Itch Numerical Rating Scale (NRS) reduction.
Rezpegaldesleukin demonstrated significant clinical efficacy. The mean EASI improvements reached up to 63% with active treatment vs approximately 29% with placebo at week 16. Notably, improvements were consistent across patients with moderate and severe disease at baseline.
Response rates increased over time. The EASI-75 and EASI-90 responses were higher with rezpegaldesleukin compared with placebo across all subgroups, including patients with severe disease.
These findings were further supported by subgroup analyses, showing consistent treatment effects regardless of baseline disease severity, geographic region, or comorbid asthma.
Safety outcomes were acceptable. While injection-site reactions were common (69.7%), they were predominantly mild to moderate (99.6%), and no increased risk of serious infections, malignancy, or conjunctivitis was observed.
“Significant improvement over placebo was observed with rezpegaldesleukin 24 μg/kg q2w in mean percent change in EASI (P<0.001), EASI-75 (P <0.001), EASI-90 (P <0.05), vIGA-AD 0/1 (P <0.05), NRS-Itch (P <0.01),” the authors concluded. “Consistent efficacy was observed irrespective of baseline AD severity for those with moderate or severe AD by EASI or vIGA-AD.”
These results position rezpegaldesleukin as a promising emerging therapy in AD, offering a novel mechanism that targets immune regulation rather than direct cytokine blockade.
Reference
Silverberg JI, Bieber T, Chovatiya R, et al. Novel regulatory T-cell enhancing biologic rezpegaldesleukin: phase 2b efficacy, safety, and baseline severity–dependent treatment response in moderate-to-severe atopic dermatitis. Presented at: American Academy of Dermatology Annual Meeting; March 27–31, 2026; Denver, Colorado.