Tildrakizumab Improves Nail Psoriasis Severity and Pain With Sustained Efficacy Through 52 Weeks
A phase 3b randomized trial demonstrates that tildrakizumab provides sustained improvements in nail psoriasis severity and associated pain in patients with moderate-to-severe plaque psoriasis affecting the nails.
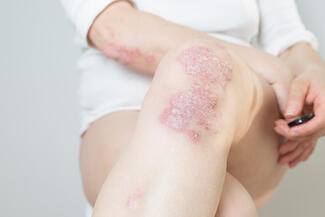
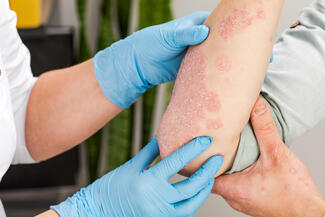
“Nail psoriasis is common and significantly impairs patients’ quality of life, but it remains challenging to manage effectively,” the investigators noted. This study evaluated the efficacy and safety of tildrakizumab over 52 weeks.
In this multicenter, double-blind, placebo-controlled trial, 99 patients were randomized to tildrakizumab 100 mg or placebo. Patients initially assigned to placebo were switched to tildrakizumab at week 28. Efficacy endpoints included ≥75% improvement in modified Nail Psoriasis Severity Index (mNAPSI 75) and ViSENPsO response, along with changes in nail pain.
Clinical responses improved over time. Also, mNAPSI 75 and ViSENPsO response rates increased from week 28 to week 52, with more than 60% of week 28 responders maintaining response at week 52. Patients who switched from placebo to tildrakizumab at week 28 demonstrated marked improvement following treatment initiation.
Nail pain also improved significantly. The mean nail pain scores decreased steadily through week 52. At week 28, a higher proportion of tildrakizumab-treated patients achieved ≥30% reduction in nail pain compared with placebo (60.6% vs 31.4%; P=0.02).
Safety findings were consistent with previous studies. No new safety signals were identified through 72 weeks, with low rates of serious adverse events and treatment discontinuation.
“The efficacy of tildrakizumab in moderate-to-severe nail psoriasis was maintained through week 52, and patients switching from placebo to tildrakizumab at week 28 showed subsequent improvement,” the authors concluded. “No new safety signals were observed through week 72.”
These findings support tildrakizumab as an effective and durable treatment option for nail psoriasis, addressing both structural disease severity and patient-reported pain.
Reference
Yamauchi PS, Gebauer K, Kerdel F, et al. Efficacy and safety of tildrakizumab in patients with moderate-to-severe psoriasis affecting the nails: a multicenter, randomized, double-blind, placebo-controlled phase 3b trial. Presented at: American Academy of Dermatology Annual Meeting; March 27–31, 2026; Denver, Colorado.