Tirbanibulin Demonstrates Significant Lesion Reduction and Favorable Safety in Actinic Keratosis Over Larger Treatment Fields
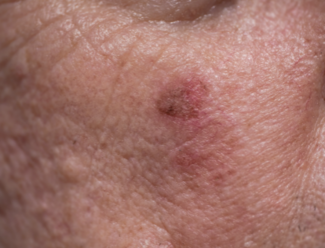
A phase 3 randomized trial demonstrates that tirbanibulin 1% ointment provides significant lesion reduction and sustained clearance in actinic keratosis (AK), even when applied to larger treatment fields up to approximately 100 cm².
In this multicenter, randomized, double-blind, vehicle-controlled phase 3 trial, 187 patients received tirbanibulin and 93 received vehicle. Patients applied treatment once daily for 5 days, with a second course administered at day 57 if complete clearance was not achieved. The primary endpoint was percent change in lesion count at day 57.
Tirbanibulin significantly reduced lesion counts compared with the vehicle. At day 57, the least-squares mean reduction was 64.2% with tirbanibulin vs 25.2% with vehicle (P<0.001), with a median reduction of 84.5% vs 20.0%. Complete clearance rates at day 113 reached 56.1% in the tirbanibulin group compared with 23.7% in the vehicle group (P<0.001).
Treatment was well tolerated. Most adverse events were mild-to-moderate local skin reactions, including erythema and flaking/scaling. These reactions peaked early (around day 8) and resolved by day 29. No new safety concerns were identified, and no squamous cell carcinoma cases were reported in the tirbanibulin group.
“At day 57, tirbanibulin showed significant and clinically meaningful efficacy and benefit up to 2 courses on fields above 25 cm² and up to approximately 100 cm²,” the authors concluded. “Safety and tolerability were consistent with previous studies… No new safety concerns [were] identified when tirbanibulin is applied in an area up to 100 cm².”
These findings support the use of tirbanibulin across larger treatment areas and reinforce its role as an effective, well-tolerated field therapy for AK.
Reference
Welzel J, Szepietowski JC, Ferrándiz Pulido C, et al. A phase 3 study assessing the efficacy, safety, and tolerability of up to two 5-day treatment courses of tirbanibulin 1% ointment in patients with actinic keratosis over a treatment field up to approximately 100 cm². Presented at: American Academy of Dermatology Annual Meeting; March 27–31, 2026; Denver, Colorado.