Cost-Effectiveness of Genicular Artery Embolization From an Interventional Cardiologist vs Interventional Radiologist Standpoint: A Model-Based Comparative Analysis
Key Summary
- A model-based cost-effectiveness analysis evaluated adults with symptomatic knee osteoarthritis using randomized controlled trial/meta-analysis inputs to compare genicular artery embolization (GAE) performed by interventional radiologists (IR) vs cardiologists (IC).
- The study found IR-performed GAE offered better value, with higher quality-adjusted life years (QALYs) and lower costs. IR-performed GAE was cost-effective in 78.4% vs IC-performed GAE in 20.6%, with ICs becoming competitive if success ≥83%, cost <$3,450, or QALY ≥0.56.
- Findings suggest outcomes are driven more by operator experience and workflow than specialty, with IC performance potentially improving with training; limitations include no direct head-to-head trials, modeled assumptions, and a 2-year horizon, supporting need for prospective and longer-term data.
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Vascular Disease Management or HMP Global, their employees, and affiliates.
VASCULAR DISEASE MANAGEMENT. 2026;23(4):E42-E53
Abstract
Background: Knee osteoarthritis is a major source of global disability, with rising prevalence and limited treatment options for patients not ready for surgery. Genicular artery embolization (GAE) has emerged as a minimally invasive alternative offering pain relief by targeting hypervascular synovial inflammation. While the clinical effectiveness of GAE is increasingly established, the economic impact of the procedure when performed by different specialties—interventional radiologists (IRs) vs interventional cardiologists (ICs)—remains unknown. Methods: A cost-effectiveness analysis was conducted using a Markov model over a 2-year horizon from the U.S. Medicare payer perspective. The model included 5 health states: controlled pain, uncontrolled pain, complication, attrition, and death. Clinical data were sourced from recent trials and meta-analyses. Costs were derived from the 2023 CMS Fee Schedule, and utility values were mapped from patient-reported outcomes. One-way sensitivity analyses, probabilistic simulations, and scenario-based threshold analyses were performed to test the robustness of the results. Results: GAE performed by IRs was the dominant strategy, yielding greater health benefits (0.58 quality-adjusted life years [QALYs]) at a lower cost ($3,940) compared to IC-performed GAE (0.51 QALYs at $4,610). In probabilistic simulations, IRs were cost-effective in 78.4% of cases at a $100,000/QALY threshold, while ICs were cost-effective in 20.6%. Scenario analyses indicated that IC-performed GAE could become cost-effective if success rates reached 83% or greater, costs dropped below $3,450, or utility gains exceeded 0.56 QALYs. Conclusion: GAE is cost-effective when performed by experienced IRs. However, ICs may deliver comparable value with improved outcomes and procedural efficiency. These findings can inform training, credentialing, and reimbursement strategies as GAE adoption expands.
Introduction
Knee osteoarthritis (KOA) is a leading cause of chronic pain and disability worldwide, with its prevalence and impact escalating rapidly.1 As of 2020, approximately 595 million individuals globally were affected by OA, representing 7.6% of the global population—a 132% increase since 1990.2 Notably, the knee is the most commonly affected joint, and cases of KOA are projected to rise by 74.9% by 2050, driven by aging populations and increasing obesity rates.2 Traditional management of KOA includes conservative therapies such as physical therapy, weight management, and pharmacologic interventions.3,4 However, these approaches often provide limited relief, especially in moderate-to-severe cases. Total knee arthroplasty (TKA) remains the definitive treatment for end-stage KOA but is associated with high costs and potential complications, and is not suitable for all patients due to age, comorbidities, or personal preferences.5-7
In this context, genicular artery embolization (GAE) has emerged as a promising minimally invasive alternative.8,9 GAE targets the hypervascular synovial membrane, contributing to inflammation and pain in KOA by embolizing the genicular arteries.10-12 Clinical studies have demonstrated that GAE can achieve significant pain relief and functional improvement. For instance, a systematic review reported that 47.4% of patients experienced a 50% or greater reduction in OA symptoms at 24 months post-procedure, with sustained benefits observed in a substantial proportion of patients.13 Despite the growing evidence supporting the efficacy of GAE, questions remain regarding the optimal provider for this procedure. Interventional radiologists (IRs) have traditionally performed GAE, while interventional cardiologists (ICs) and vascular surgeons, with extensive experience in peripheral endovascular interventions, are increasingly adopting this technique.14,15 ICs, with their proficiency in catheter-based techniques, are increasingly adopting GAE into their practice.16,17 This shift raises important considerations about the comparative effectiveness and cost-efficiency of GAE when performed by different specialists.18
Economic evaluations are crucial in this context as healthcare systems strive to allocate resources efficiently.19,20 A recent cost-effectiveness analysis indicated that GAE is more cost-effective than other nonsurgical interventions, such as radiofrequency ablation and corticosteroid injections, particularly when clinical success rates exceed 51% and utility values exceed 0.617. 21,22 However, these analyses have not differentiated outcomes based on the provider's specialty, leaving a gap in understanding the economic implications of GAE performed by IRs vs ICs.
This study aims to address this gap by conducting a model-based cost-effectiveness analysis comparing GAE performed by IRs and ICs for the treatment of symptomatic KOA. Utilizing a validated Markov model informed by clinical trial data, real-world cost inputs, and utility estimates derived from patient-reported outcomes, we evaluate the economic and clinical value of GAE across provider specialties. Our goal is to inform clinical practice, training programs, and policy decisions to optimize patient outcomes and resource utilization in the management of KOA.
Importantly, this analysis is not intended to compare medical specialties as intrinsic determinants of procedural quality. Rather, operator specialty is used as a pragmatic surrogate for differences in procedural maturity, case volume, workflow optimization, and early learning-curve effects observed during the diffusion of new endovascular therapies. Outcomes following GAE are fundamentally driven by technical completeness, experience, and delivery setting, irrespective of training background. Accordingly, this study evaluates how differences in procedural adoption and system-level factors may influence the economic value of GAE.
Methods
Study design and perspective
This study employed a model-based cost-effectiveness analysis to evaluate GAE performed by IRs vs ICs for the treatment of symptomatic KOA. The analysis was conducted from the perspective of a U.S. Medicare healthcare payer, using a time horizon of 2 years.23. A state-transition (Markov) decision model was developed in Microsoft Excel to simulate clinical and economic outcomes, incorporating real-world clinical trial data, procedural costs, health utility estimates, and transition probabilities. This study involved no patient-level data and was therefore exempt from institutional review board approval.
Data sources and literature selection
To populate the Markov model with clinically and economically meaningful inputs, we conducted a structured literature review from January 2010 to February 2025. Our search focused on peer-reviewed clinical trials, meta-analyses, and economic evaluations related to GAE for KOA. Databases searched included PubMed, Embase, Cochrane Library, and Scopus, using the following keywords: “genicular artery embolization”, “knee osteoarthritis”, “GAE outcomes”, “interventional radiology”, “interventional cardiology”, “cost-effectiveness”, “quality of life”, “QALY”, “VAS”, “WOMAC”, and “utility mapping”. Only English-language studies with adult populations (≥18 years) and a minimum follow-up of 6 months were included.
Clinical effectiveness data were primarily sourced from 3 high-quality randomized controlled trials (RCTs) (Landers et al 2023, van Zadelhoff et al 2024, Bagla et al 2022) and the meta-analysis by Taslakian et al (2023), which provided pooled estimates on pain reduction, functional improvement, and success rates.10,24-26 Utility values were mapped from validated Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) and Visual Analog Scale (VAS) score conversions using established methods from the OA literature.
Cost inputs were derived from the 2023 U.S. Centers for Medicare & Medicaid Services (CMS) Physician Fee Schedule, supplemented with institutional data and cost-effectiveness studies on other KOA interventions (eg, corticosteroid injection, radiofrequency ablation).27 Transition probabilities were synthesized from prospective cohort studies and pooled where needed. Only sources with clear methodology, defined endpoints, and adequate sample sizes (≥30 patients) were considered for model inputs.28
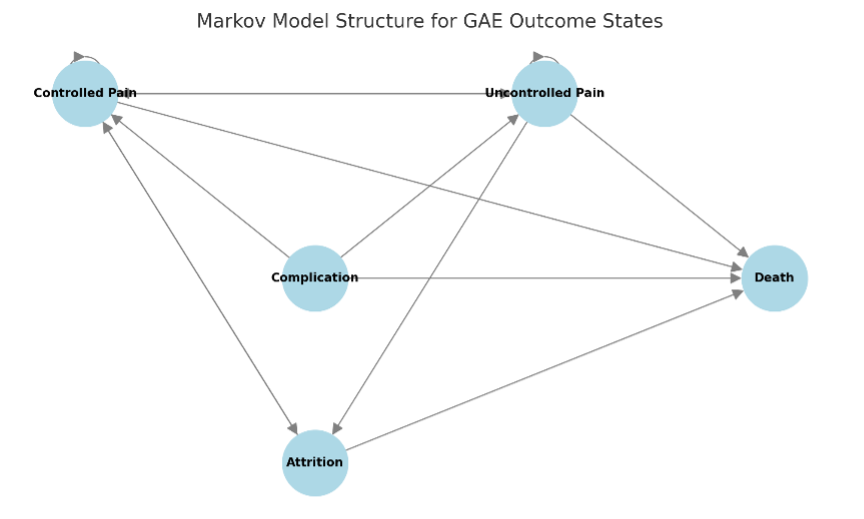
Model structure
The Markov model comprised 5 mutually exclusive health states: controlled pain (representing successful GAE), uncontrolled pain (representing partial or failed response), complication (reflecting procedure-related adverse events), attrition (loss to follow-up, dropout, or treatment crossover), and death (absorbing state). These health states were selected to represent clinically meaningful and mutually exclusive outcomes of GAE over time and are consistent with prior economic models used in musculoskeletal interventions and decision-analytic literature.24,28 Patients entered the model following GAE and could transition between states in eight 3-month cycles over the 2-year period. Transition probabilities between health states were derived from RCTs, systematic reviews, and prospective cohort studies. All outcomes were discounted at an annual rate of 3%, and half-cycle correction was applied in accordance with health economic modeling standards.
Mathematical model and economic evaluation
Cost-effectiveness was evaluated by calculating the incremental cost-effectiveness ratio (ICER), a standard metric in health economic evaluations that quantifies the additional cost per additional quality-adjusted life year (QALY) gained. The ICER was computed using the following equation:
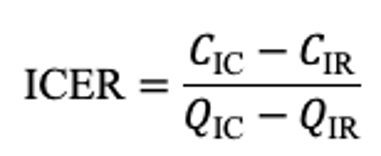
where CIC and CIR represent the total costs of GAE when performed by ICs and IRs, respectively, and QIC and QIR denote the corresponding QALYs gained over the 2-year horizon. To calculate total QALYs accrued over time, each patient’s utility value in a given health state was multiplied by the duration spent in that state and adjusted for temporal discounting. The following formula was applied across all 8 cycles in the Markov model:
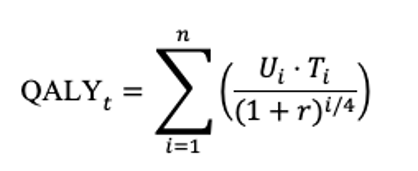
In this equation, Ui represents the utility value assigned to the patient’s health state during the ith cycle, and Ti denotes the duration of each Markov cycle, fixed at 0.25 years (3 months). The term r corresponds to the annual discount rate, set at 3%, while iii refers to the cycle number, ranging from 1 to 8 over the 2-year model horizon.
The expression
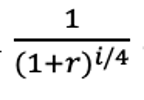
applies quarterly discounting with half-cycle correction to reflect the present value of utility accrued during each time interval. All costs were modeled in 2023 U.S. dollars from the perspective of a Medicare healthcare payer. Discounting was applied symmetrically to both costs and QALYs. Outcomes were reported as total QALYs, total costs, and ICERs, with dominance defined as a strategy being both more effective and less costly than the comparator (Figure 1).
Utility values and health state assignment
Health utility weights for each health state were assigned based on validated mappings from these clinical outcomes, using standard values from the OA literature. For successful GAE, the utility value was set at 0.75 for IR and 0.70 for IC, reflecting differences in procedural success based on the published learning curve data. The utility for uncontrolled pain was set at 0.50 (IR) and 0.45 (IC), while complication and attrition states were assigned a utility of 0.30 and 0.10, respectively. Death was assigned a utility of 0.00. These values were applied to each cycle to compute QALYs accrued over the 2-year model period.
Cost estimation
Cost estimates were primarily derived from the 2023 CMS Physician Fee Schedule and supplemented with published cost-effectiveness studies and real-world procedural data. The procedural cost of GAE performed by IRs was estimated at $3100, reflecting outpatient interventional suite delivery with streamlined radiology-specific workflows. In contrast, GAE performed by ICs was modeled at $3650, based on reasonable assumptions of higher facility utilization, longer procedural time, and potentially less optimized scheduling infrastructure, as ICs represent newer adopters of this procedure. Follow-up care costs, including medication, imaging, and physical therapy over a 2-year period, were standardized at $500 for both arms to maintain comparability. Attrition-related costs—representing retreatment, escalation to surgery, or clinical dropout—were estimated at $340 for IRs and $460 for ICs, based on higher attrition probabilities observed in trials and early-phase implementation data for newer operators. These cost assumptions were conservative, literature-informed, and subjected to rigorous one-way and probabilistic sensitivity analyses to evaluate their impact on model outcomes. Total modeled per-patient costs amounted to $3940 for IR-performed GAE and $4610 for IC-performed GAE.
Transition probabilities
Transition probabilities were informed by pooled clinical data. The procedural success rate was estimated at 85% for IR and 75% for IC, with technical success rates near 100% for both based on multiple cohort studies. Attrition was assumed to be 10% for IR and 15% for IC over the 2-year period. The rate of complications was low in all reviewed trials and meta-analyses (<2%) and was assumed to be equal across both arms. Mortality rates were modeled according to age-specific background mortality data and did not differ between arms.
Deterministic sensitivity analysis
To assess the robustness of the model, one-way deterministic sensitivity analysis was performed. Key model parameters, including procedural cost, clinical success rate, utility gain, and attrition rate, were varied across plausible ranges derived from the literature. The resulting changes in the ICER were evaluated, and a tornado diagram was generated to illustrate which variables had the greatest influence on model outcomes.
Threshold and scenario analyses
A threshold analysis was conducted to identify critical breakpoints at which IC-performed GAE would become cost-effective under a willingness-to-pay (WTP) threshold of $100,000 per QALY. In parallel, several scenario analyses were developed to simulate real-world conditions: one scenario assumed equal clinical effectiveness for IR and IC; another equalized procedural costs between specialties; and a third modeled both procedures being conducted in office-based labs to reduce facility overhead.
Probabilistic sensitivity analysis
To account for joint uncertainty across all parameters, a probabilistic sensitivity analysis (PSA) was conducted using a second-order Monte Carlo simulation with 10,000 iterations. Probability distributions were assigned to all uncertain parameters: gamma distributions for cost variables and beta distributions for utilities and probabilities. Simulation results were used to construct cost-effectiveness acceptability curves (CEACs) and estimate the probability that each strategy would be considered cost-effective at various WTP thresholds, including $50,000, $100,000, and $150,000 per QALY.
Outcome measures
The primary outcome measure was the ICER, calculated as the difference in total costs divided by the difference in QALYs between IC and IR strategies. Secondary outcomes included net monetary benefit and the probability of cost-effectiveness based on WTP thresholds.
Key model assumptions
Several assumptions were made to ensure model tractability and policy relevance. First, complication and mortality rates were assumed to be equivalent between groups, reflecting the low incidence of serious adverse events reported across published trials. Second, operator specialty was modeled as a proxy for procedural experience, workflow efficiency, and embolization completeness, rather than as an independent causal determinant of outcomes. Third, health utilities were derived through validated mappings from WOMAC and VAS scores rather than direct utility elicitation instruments. Finally, a 2-year time horizon was selected to reflect the current strength of clinical durability data for GAE while avoiding speculative long-term extrapolation.
Vascular surgeons were not modeled as a separate comparator arm due to the absence of specialty-stratified outcome data; however, their established expertise in peripheral arterial anatomy and embolization-adjacent techniques positions them within the same procedural maturity spectrum as experienced endovascular operators.
Results
Base case cost-effectiveness results
In the base case analysis, GAE performed by IRs yielded superior economic and health outcomes compared to GAE performed by ICs. Over a 2-year time horizon, the total cost of IR-performed GAE was estimated at $3940, while IC-performed GAE incurred a higher cost of $4610. In terms of health outcomes, the IR arm provided a utility gain of 0.58 QALYs compared to 0.51 QALYs in the IC arm.
Because the IR strategy resulted in both lower costs and greater effectiveness, it was considered the dominant strategy in the base case scenario. As such, the ICER was not calculated, since the comparator (IC arm) was both more costly and less effective (Table 1).
Table 1. Base case cost-effectiveness results.
| Strategy | Total cost (USD) | QALYs gained | ICER vs next best |
| GAE by interventional radiologist | $3940 | 0.58 | Dominant |
| GAE by interventional cardiologist | $4610 | 0.51 | - |
Abbreviations: USD, U.S. dollars; QALYs, quality-adjusted life years; ICER, incremental cost-effectiveness ratio; GAE, genicular artery embolization.
The cost difference primarily reflected variations in procedural setting and efficiency. IRs were assumed to perform GAE in outpatient interventional radiology suites with optimized workflows, while IC-performed GAE was modeled with higher institutional overhead and modestly increased procedure times. The observed effectiveness differential was informed by published RCTs, which indicated a learning curve for operators early in their experience with GAE (Figure 2).
Clinical effectiveness and utility gains
The most recent meta-analysis by Taslakian et al (2023) demonstrated that GAE achieved a mean reduction of 34 to 39 points on the VAS for pain and an improvement of 28 to 34 points in WOMAC scores at 6 to12 months post-procedure. These changes exceed the minimum clinically important difference (MCID), indicating substantial clinical benefit. Moreover, at 1 year, 92% of patients met MCID criteria for WOMAC, and 78% achieved substantial clinical benefit thresholds.
These outcomes were mapped to utility values to estimate QALYs. Successful GAE was assigned a utility value of 0.75 in the IR arm and 0.70 in the IC arm, with lower utility scores allocated for suboptimal outcomes. The slight decrement for the IC arm reflects operator variability and technical completeness as reported in early-phase trials. Utilities for other health states were applied equally across arms, based on literature-standard values. These utility estimates, applied across eight 3-month cycles, yielded cumulative QALYs of 0.58 for IR-performed GAE and 0.51 for IC-performed GAE over the model horizon (Table 2).
Table 2. Utility values by health state and operator type.
| Health state | Utility (IR) | Utility (IC) |
| Controlled pain (successful GAE) | 0.75 | 0.70 |
| Uncontrolled pain | 0.50 | 0.45 |
| Complication | 0.30 | 0.30 |
| Attrition | 0.10 | 0.10 |
| Death | 0.00 | 0.00 |
Abbreviations: IR, interventional radiologist; IC, interventional cardiologist; GAE, genicular artery embolization.
Cost components and resource utilization
Cost components were estimated based on the 2023 Medicare Physician Fee Schedule, literature on outpatient interventional care, and trial-based follow-up data. The procedural cost of GAE was estimated at $3100 when performed by IRs in an outpatient setting. For ICs, the cost was modeled at $3650, accounting for potential use of hospital-based cardiac catheterization laboratories and longer procedure times associated with learning curves. Follow-up care, including routine visits, physical therapy referrals, and imaging, was assumed equal between the two groups at $500 per patient.
Attrition management, reflecting additional treatments for non-responders (eg, repeat injections or surgical consultation), was modeled at $340 in the IR arm and $460 in the IC arm, based on higher observed attrition in IC-related outcome data (Table 3).
Table 3. Cost breakdown by operator type.
| Cost component | IR-performed GAE | IC-performed GAE |
| Procedural cost | $3100 | $3650 |
| Follow-up care (2 years) | $500 | $500 |
| Attrition management | $340 | $460 |
| Total cost | $3940 | $4610 |
Abbreviations: IR, interventional radiologist; GAE, genicular artery embolization; IC, interventional cardiologist.
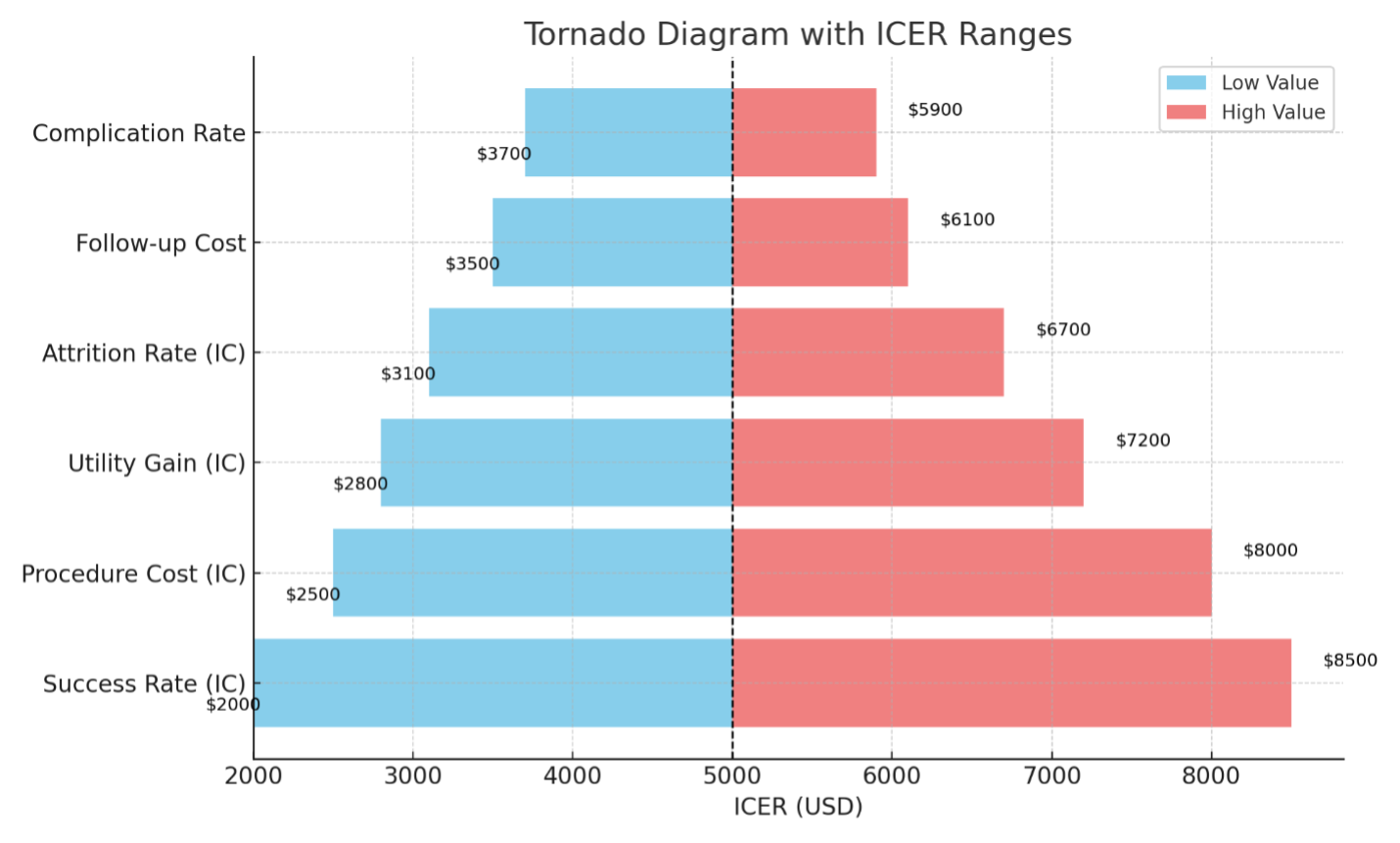
Deterministic sensitivity analysis
One-way sensitivity analysis was conducted to identify the variables with the greatest influence on cost-effectiveness outcomes. The ICER was most sensitive to changes in the clinical success rate of IC-performed GAE, followed by procedural cost and utility gain.
The analysis demonstrated that IC-performed GAE could become cost-effective under a WTP threshold of $100,000 per QALY if the success rate reached ≥83%, the utility gain exceeded 0.56 QALYs, or if the total procedural cost fell below $3,450. These thresholds represent plausible targets that could be attained with enhanced training and outpatient procedural delivery models.
Probabilistic sensitivity analysis
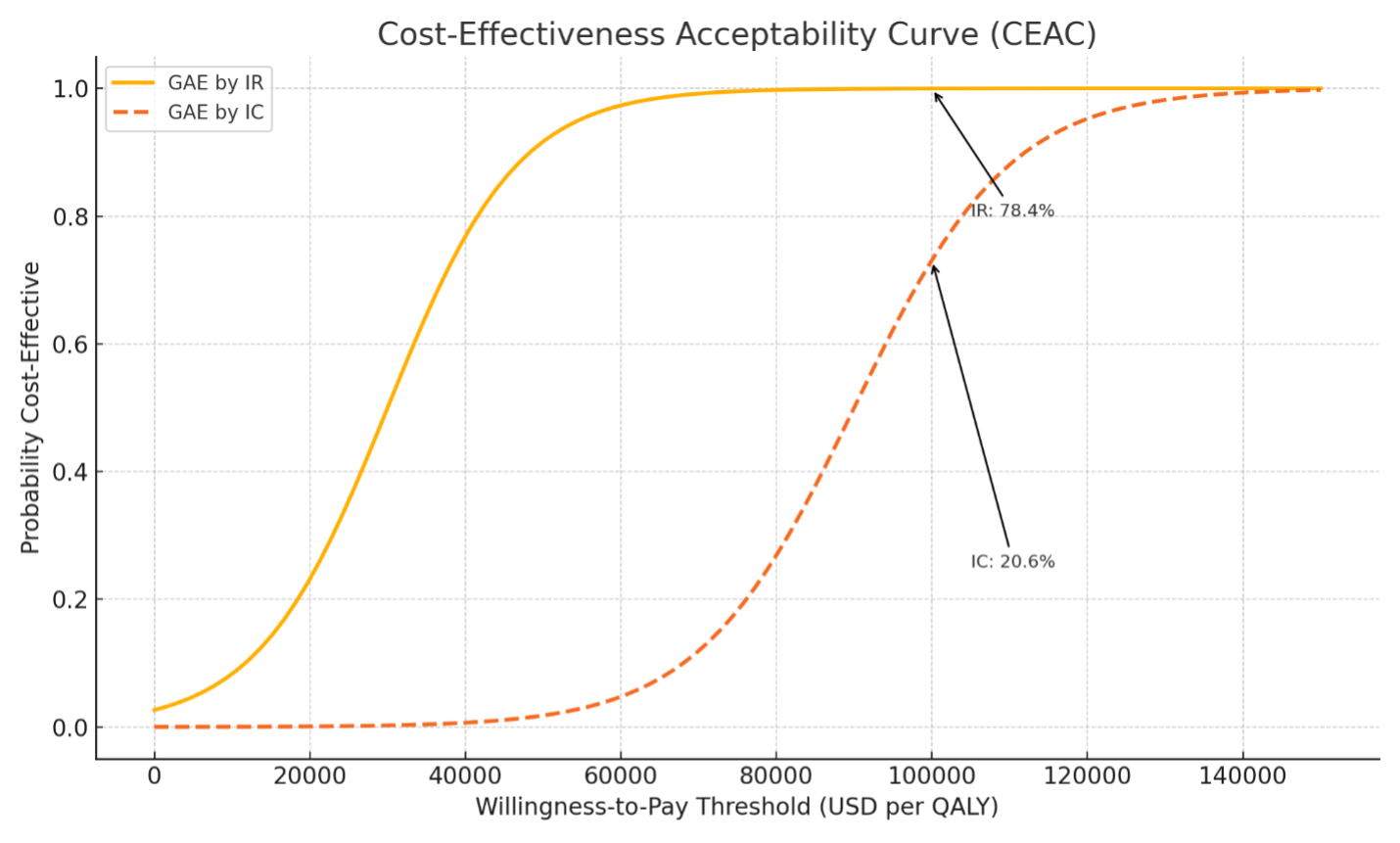
A PSA was performed using 10,000 Monte Carlo simulations. Probability distributions were applied to all uncertain parameters: gamma distributions for costs and beta distributions for utilities and success rates. The outcomes were plotted as a CEAC, revealing the probability that each strategy would be considered cost-effective at varying WTP thresholds (Figure 3).
At a WTP of $100,000 per QALY, GAE performed by IRs was cost-effective in 78.4% of simulations, compared to 20.6% for the IC strategy. The remaining simulations reflected overlapping uncertainty or dominated scenarios (Table 4).
Table 4. Mean probabilistic sensitivity analysis.
| Strategy | Mean cost (USD) | Mean QALYs | Probability cost-effective |
| GAE by interventional radiologist | $3955 | 0.576 | 78.4% |
| GAE by interventional cardiologist | $4625 | 0.507 | 20.6% |
Abbreviations: QALY, quality-adjusted life years; GAE, genicular artery embolization.
Scenario and threshold analyses
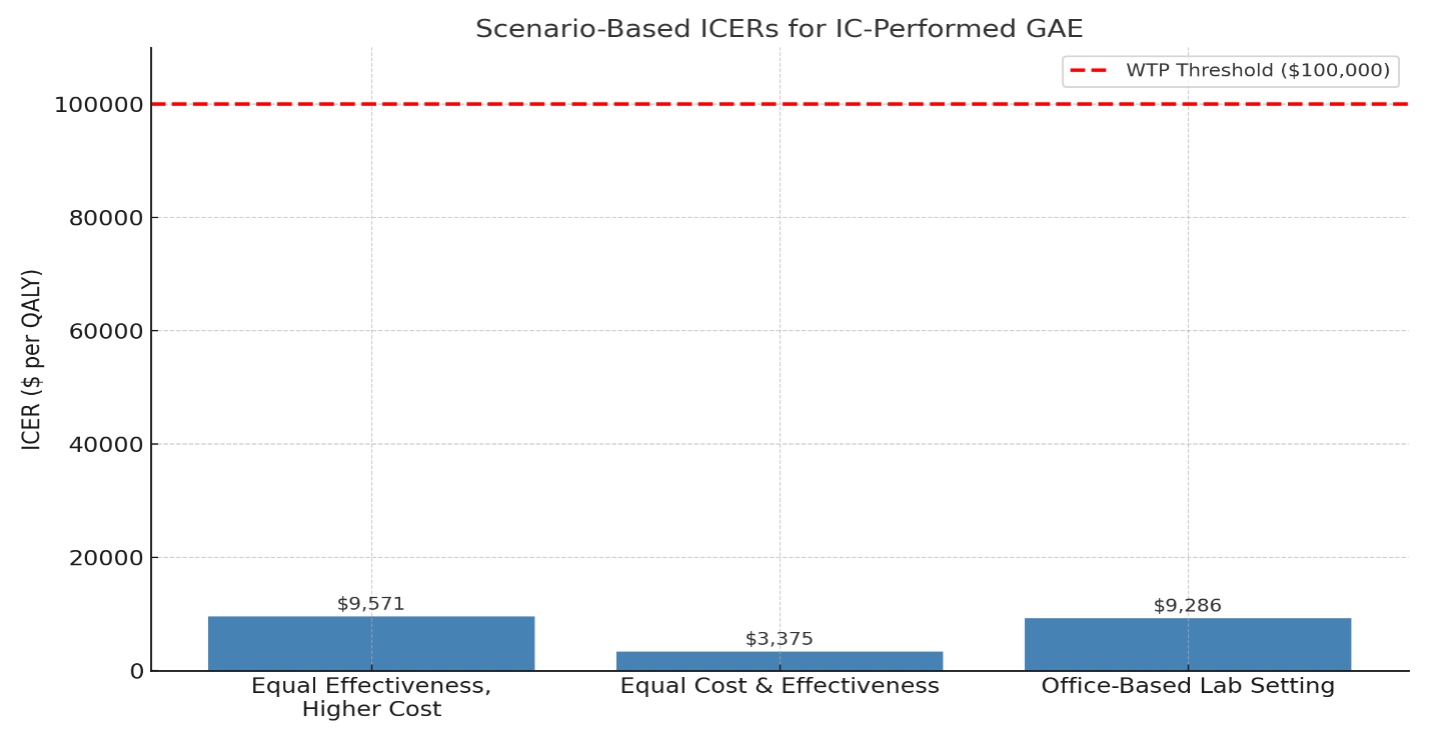
Several scenario analyses were conducted to simulate real-world variations and test the feasibility of IC-performed GAE achieving cost-effectiveness. In a scenario where both IR and IC achieved equal clinical effectiveness (85% success rate, 0.58 QALYs), but the IC arm retained a higher procedural cost, the resulting ICER for IC was $9571 per QALY, well below the WTP threshold.
In another scenario, procedural costs for ICs were reduced to match IRs ($3100), with effectiveness held constant. Under these conditions, the ICER dropped further to $3375 per QALY, indicating strong cost-effectiveness even at current performance levels.
A third scenario explored GAE performed in office-based labs by both specialties, reducing procedural cost to $2500. In this model, IR-performed GAE remained dominant, though IC-performed GAE also became highly cost-effective with an ICER of $9286 per QALY.
Threshold analyses indicated that IC-performed GAE would become cost-effective if any one of the following were true: the success rate reached 83% or higher, utility gain 0.56 QALYs or higher, or procedural cost $3450 or less. These represent reasonable benchmarks for training and practice optimization (Figure 4).
Discussion
In this model-based cost-effectiveness analysis, GAE performed within a mature procedural delivery framework was economically dominant, yielding higher health benefits at lower cost. When modeled using IRs as the reference group for procedural maturity and system efficiency, GAE demonstrated superior value compared with less-optimized adoption scenarios represented by ICs in early-phase implementation.
The cost-effectiveness advantage of IR-performed GAE is largely attributable to procedural maturity and efficiency. IRs have played a leading role in the development and evolution of embolization techniques across various organ systems, and they often operate in streamlined outpatient interventional suites. This experience likely contributes to higher rates of complete embolization, shorter procedure times, and lower facility-related costs. In contrast, ICs—while skilled in endovascular navigation—are relatively new to musculoskeletal embolotherapy. The procedural learning curve for ICs was evident in post hoc subgroup analyses from the 2023 randomized trial by Landers et al, which found that complete embolization was a significant determinant of pain relief and that early cases performed with partial embolization were less clinically effective.24
Our findings align with and extend those of prior studies. Kwak et al (2024) previously demonstrated that GAE was more cost-effective than genicular nerve radiofrequency ablation or corticosteroid injection, with ICERs well below standard WTP thresholds.22 Their model, however, did not distinguish operator types. By introducing this variable, our study provides novel insight into how procedural delivery and experience shape the economic value of minimally invasive therapies. Our results also echo the conclusions of the Spanish Health Technology Assessment (2023), which estimated that GAE would be cost-effective if it produced even modest QALY gains—gains that our analysis confirms are achievable with IRs and potentially with ICs as procedural experience grows.8
Importantly, our model did not suggest that IC-performed GAE is intrinsically nonviable. In fact, in scenarios where ICs achieved comparable success rates to IRs or delivered GAE in lower-cost office-based settings, IC-performed GAE became cost-effective with ICERs below $10,000 per QALY. These thresholds are not hypothetical ideals; they are realistic targets that could be met through structured training, interdisciplinary collaboration, and shifts in procedural delivery. As such, our findings support a vision in which ICs can become integral providers of GAE, provided that clinical standards are met and economic efficiencies are optimized.29
Vascular surgeons represent an additional and often underrecognized stakeholder in the delivery of GAE. Given their extensive experience with peripheral arterial disease, microcatheter techniques, and limb-specific vascular anatomy, vascular surgeons are well positioned to perform GAE within appropriately structured endovascular programs. In many health systems, vascular surgery already operates within high-volume, cost-efficient procedural environments comparable to IR suites, suggesting that outcomes and economic performance may more closely resemble mature IR workflows than early-phase adoption scenarios.
Taken together, these findings support a multidisciplinary model of GAE delivery involving IRs, vascular surgeons, and ICs, with value determined primarily by operator experience, procedural standardization, and delivery setting rather than specialty designation. Such a framework aligns with contemporary trends in endovascular care, where procedural competency is increasingly defined by volume, outcomes, and infrastructure rather than traditional specialty boundaries.
These findings carry important implications for the diffusion of GAE into broader clinical practice. Premature expansion of GAE without standardized training pathways, procedural benchmarks, and volume-based competency thresholds may result in heterogeneous outcomes that undermine payer confidence and reimbursement stability. Early variability in effectiveness, rather than the therapy itself, poses the greatest risk to long-term adoption.
Conversely, our scenario and threshold analyses demonstrate that IC-performed GAE is not intrinsically disadvantaged. When procedural success rates, embolization completeness, and delivery costs approximate those of established IR workflows, IC-performed GAE achieves strong cost-effectiveness well below conventional WTP thresholds. These findings support structured cross-specialty training, interdisciplinary collaboration, and registry-based outcome monitoring as mechanisms to ensure value preservation during procedural diffusion.
From a clinical perspective, GAE represents a valuable therapeutic option for patients with moderate KOA who are not yet candidates for TKA or wish to delay surgery. RCTs and observational studies have consistently shown significant pain and function improvements, with favorable safety profiles and durable outcomes extending to 2 years.30-32 Our analysis adds a critical dimension to that evidence: not only is GAE clinically beneficial, but when performed by experienced providers, it also represents a high-value intervention from a healthcare system perspective. Ultimately, this study provides a timely economic framework to support credentialing decisions, training investments, and reimbursement models in the evolving landscape of image-guided musculoskeletal interventions. As the role of multidisciplinary endovascular care continues to grow, it will be essential to balance innovation with value, ensuring that new procedural ownership models maintain both clinical excellence and economic sustainability.
Limitations and Strengths
This study has several limitations that warrant consideration. First, while our model differentiates GAE performance by IRs and ICs, no randomized or observational studies currently offer a direct head-to-head comparison by operator specialty. Additionally, the absence of specialty-specific outcome data for vascular surgeons precluded their inclusion as a separate comparator, highlighting an important area for future registry-based and comparative research.
As such, our assumptions regarding success rates, utility gains, and cost differences between IRs and ICs were based on post hoc subgroup analyses, procedural literature, and practice-based modeling rather than stratified empirical data. These assumptions, while evidence-informed, introduce a degree of uncertainty.
Second, health utility values were derived from patient-reported outcomes (eg, VAS and WOMAC) mapped to QALY estimates using established algorithms, rather than from direct utility elicitation methods (eg, EuroQol-5 Dimensions or Short-Form 6-Dimension Health Utility Index). Although this approach is widely accepted in musculoskeletal modeling, it may introduce approximation error or oversimplify the patient experience. Third, the cost data were modeled from a U.S. CMS perspective and may not reflect costs or reimbursement patterns in other countries or private-payer systems. Variables such as institutional pricing, provider fees, and access to office-based labs could substantially impact real-world generalizability.
Additionally, while our model included a broad range of sensitivity analyses and probabilistic simulations, the long-term durability of GAE remains an evolving area of evidence. While cohort studies suggest sustained benefit over 1 to 2 years, future data may modify assumptions on retreatment rates or utility decay over time. Similarly, ICs adopting GAE are still in the early phase of implementation, and future improvements in technique or cost structure may shift outcomes in their favor beyond what current data suggest.
Despite these limitations, this study has several notable strengths. It is the first economic analysis to compare GAE by operator specialty, using a validated Markov model, real-world clinical effectiveness data from randomized trials and meta-analyses, and cost inputs from authoritative sources such as the CMS Fee Schedule. Our modeling approach followed best practices, including half-cycle correction, PSA, and scenario modeling to reflect both ideal and real-world conditions. By integrating both clinical effectiveness and procedural dynamics into a single framework, we provide an evidence-based foundation for institutional policymaking, training programs, and reimbursement considerations.
Conclusion
In this model-based cost-effectiveness analysis, we found that GAE performed by IRs is the dominant strategy for the treatment of symptomatic KOA, delivering higher health benefits at a lower cost when compared to GAE performed by ICs. Specifically, IR-performed GAE was associated with a lower total cost ($3940 vs $4610) and higher QALY gain (0.58 vs 0.51) over a 2-year time horizon. These findings were consistent across deterministic and probabilistic sensitivity analyses, with IRs maintaining cost-effectiveness in over 78% of modeled scenarios.
However, our scenario and threshold analyses demonstrated that IC-performed GAE can become cost-effective if key performance parameters are optimized. When success rates increased or costs decreased, such as through experience gain, structured training, or office-based procedural settings, ICs achieved ICERs well below commonly accepted WTP thresholds.
In conclusion, GAE, when performed within established, high-efficiency procedural frameworks, is a high-value intervention for symptomatic KOA. While IR-performed GAE was economically dominant in the base case, IC-performed GAE achieved cost-effectiveness under clearly defined performance and cost thresholds. These results highlight that operator experience, procedural standardization, and delivery setting, across interventional radiology, vascular surgery, and interventional cardiology, ultimately determine the economic value of GAE.
As GAE adoption expands, aligning training, credentialing, and reimbursement with these principles will be essential to sustaining both clinical and economic benefit.
Affiliations and Disclosures
Darshan Hullon, DO, is from MercyOne Clinton Medical Center, Clinton, Iowa and Abiya Ahad, MBBS, is from the Ananta Institute of Medical Sciences and Research Centre, Udaipur, India.
The authors report no financial relationships or conflicts of interest regarding the content herein.
Manuscript accepted March 12, 2026.
Address for correspondence: Abiya Ahad, MBBS, Ananta Institute of Medical Sciences and Research Centre, Udaipur, Rajasthan, India. Email: ahadabiya15@gmail.com
References
1. Geng R, Li J, Yu C, et al. Knee osteoarthritis: current status and research progress in treatment (Review). Exp Ther Med. 2023;26(4):481. doi:10.3892/etm.2023.12180
2. GBD 2021 Osteoarthritis Collaborators. Global, regional, and national burden of osteoarthritis, 1990–2020 and projections to 2050: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Rheumatol. 2023;5(9):e508-e522. doi:10.1016/S2665-9913(23)00163-7
3. Lim WB, Al-Dadah O. Conservative treatment of knee osteoarthritis: a review of the literature. World J Orthop. 2022:13(3):212-229. doi:10.5312/wjo.v13.i3.212
4. Ferreira RM, Martins PN, Gonçalves RS. Non-pharmacological and non-surgical interventions to manage patients with knee osteoarthritis: an umbrella review 5-year update. Osteoarthr Cartil Open. 2024;6(3):100497. doi:10.1016/j.ocarto.2024.100497
5. Steinhaus ME, Christ AB, Cross MB. Total knee arthroplasty for knee osteoarthritis: support for a foregone conclusion? HSS J. 2017;13(2):207-210. doi:10.1007/s11420-017-9558-4
6. Medical Clinical Policy Bulletins: Knee Arthroplasty. Aetna. https://www.aetna.com/cpb/medical/data/600_699/0660.html
7. Feng JE, Novikov D, Anoushiravani AA, Schwarzkopf R. Total knee arthroplasty: improving outcomes with a multidisciplinary approach. J Multidiscip Healthc. 2018;11:63-73. doi:10.2147/JMDH.S140550
8. Hernández-Yumar A, González-Hernández Y, Del Pino-Sedeño T, et al. Genicular artery embolization for knee osteoarthritis: a systematic review with meta-analysis and cost-analysis. Gac Sanit. 2025:39:102459. doi:10.1016/j.gaceta.2025.102459
9. Genicular Artery Embolization: A Comprehensive Insight | Advanced Vascular Centers. https://advancedvascularcenters.com/genicular-artery-embolization-technique-a-comprehensive-insight
10. Padia SA, Genshaft S, Blumstein G, et al. Genicular artery embolization for the treatment of symptomatic knee osteoarthritis. JB JS Open Access. 2021;6(4):e21.00085. doi:10.2106/JBJS.OA.21.00085
11. Taslakian B, Miller LE, Mabud TS, et al. Genicular artery embolization for treatment of knee osteoarthritis pain: systematic review and meta-analysis. Osteoarthr Cartil Open. 2023;5(2):100342. doi:10.1016/j.ocarto.2023.100342
12. Brown JM, Vandeveer ZT, Cadoret D, Morrison JJ, Jahangiri Y. Genicular artery embolization: a technical review of anatomy, pathophysiology, current experiences, and future directions. J Clin Med. 2025;14(6):2106. doi:10.3390/jcm14062106
13. Charlesworth J, Fitzpatrick J, Perera NKP, Orchard J. Osteoarthritis-a systematic review of long-term safety implications for osteoarthritis of the knee. BMC Musculoskelet Disord. 2019;20(1):151. doi:10.1186/s12891-019-2525-0
14. Singh A, Kumar A, Kumar P, Kumar S, Gamanagatti S. “Beyond saving lives”: current perspectives of interventional radiology in trauma. World J Radiol. 2017:9(4):155-177. doi:10.4329/wjr.v9.i4.155
15. Maingard J, Kok HK, Ranatunga D, et al. The future of interventional and neurointerventional radiology: learning lessons from the past. Br J Radiol. 2017;90(1080):20170473. doi:10.1259/bjr.20170473
16. Writing Committee; Bass TA, Abbott JD, et al. 2023 ACC/AHA/SCAI advanced training statement on interventional cardiology (coronary, peripheral vascular, and structural heart interventions): a report of the ACC Competency Management Committee. J Soc Cardiovasc Angiogr Interv. 2023;2(2):100575. doi:10.1016/j.jscai.2022.100575
17. Vallabhajosyula S, Alasnag M, Boudoulas KD, et al; Critical Care Cardiology Working Group of the American College of Cardiology Interventional Section Leadership Council. Future training pathways in percutaneous coronary interventions: interventional critical care, complex coronary interventions, and interventional heart failure. JACC Adv. 2024;3(11):101338. doi:10.1016/j.jacadv.2024.101338
18. Prinja S, Nandi A, Horton S, Levin C, Laxminarayan R. Costs, effectiveness, and cost-effectiveness of selected surgical procedures and platforms. In: Debas HT et al, eds. Essential Surgery: Disease Control Priorities, Third Edition, Volume 1. The International Bank for Reconstruction and Development/The World Bank; 2015.
19. Mosadeghrad AM, Jaafaripooyan E, Zamandi M. Economic evaluation of health interventions: a critical review. Iran J Public Health. 2022:51(10):2159-2170. doi:10.18502/ijph.v51i10.10975
20. Turner HC, Archer RA, Downey LE, et al. An introduction to the main types of economic evaluations used for informing priority setting and resource allocation in healthcare: key features, uses, and limitations. Front Public Health. 2021;9:722927. doi:10.3389/fpubh.2021.722927
21. AuntMinnie.com. GAE could be cost-effective for knee osteoarthritis. 2024. https://www.auntminnie.com/clinical-news/interventional/article/15704384/gae-could-be-costeffective-for-knee-osteoarthritis.
22. Kwak DH, Hofmann HL, Patel M, et al. Genicular artery embolization, radiofrequency ablation, and corticosteroid therapy for knee osteoarthritis: a cost-effectiveness analysis using randomized clinical trial data. AJR Am J Roentgenol. 2024;223(6):e2431710. doi:10.2214/AJR.24.31710
23. Kim DD, Wilkinson CL, Pope EF, Chambers JD, Cohen JT, Neumann PJ. The influence of time horizon on results of cost-effectiveness analyses. Expert Rev Pharmacoecon Outcomes Res. 2017;17(6):615-623. doi:10.1080/14737167.2017.1331432
24. Landers S, Helly R, Hely A, et al. Genicular artery embolization for early-stage knee osteoarthritis: results from a triple-blind single-centre randomized controlled trial. Bone Jt Open. 2023;4(3):158-167. doi:10.1302/2633-1462.43.BJO-2022-0161.R2
25. Van Zadelhoff TA, Bos PK, Moelker A, Bierma-Zeinstra SMA, van der Beijden RA, Oei EHG. Genicular artery embolisation versus sham embolisation for symptomatic osteoarthritis of the knee: a randomised controlled trial. BMJ Open. 2924;14(10):e087047. doi:10.1136/bmjopen-2024-087047
26. Bagla S, Piechowiak R, Sajan A, Orlando J, Hartman T, Isaacson A. Multicenter randomized sham controlled study of genicular artery embolization for knee pain secondary to osteoarthritis. J Vasc Interv Radiol. 2022;33(1):2-10.e2. doi:10.1016/j.jvir.2021.09.019
27. Centers for Medicare and Medicaid Services. Calendar Year (CY) 2023 Medicare Physician Fee Schedule Final Rule | CMS. https://www.cms.gov/newsroom/fact-sheets/calendar-year-cy-2023-medicare-physician-fee-schedule-final-rule.
28. Gidwani R, Russell LB. Estimating transition probabilities from published evidence: a tutorial for decision modelers. Pharmacoeconomics. 2020;38(11):1153-1164. doi:10.1007/s40273-020-00937-z
29. Gavaldà-Espelta E, Del Mar Lleixà-Fortuño M, Baucells-Lluis J, et al. Innovative health and social integrated care model effectiveness to improve quality care for chronic patients: a single group assignment clinical trial. Int J Integr Care. 2023:23(4):2. doi:10.5334/ijic.6759.
30. UCLA Health. Genicular Artery Embolization (GAE) - Interventional Radiology. https://www.uclahealth.org/medical-services/radiology/interventional-radiology/treatments-procedures/genicular-artery-embolization-gae.
31. O’Brien S, Blakeney WG, Soares J. Selective genicular artery embolization in the management of osteoarthritic knee pain—a narrative review. J Clin Med. 2024;13(11):3256. doi:10.3390/jcm13113256
32. Epelboym Y, Mandell JC, Collins JE, et al. Genicular artery embolization as a treatment for osteoarthritis related knee pain: a systematic review and meta-analysis. Cardiovasc Intervent Radiol. 2023;46(6):760-769. doi:10.1007/s00270-023-03422-0













