Tailored Solutions for Structural Valve Degeneration: Valve-in-Valve-in-Valve TAVR With Coronary Leaflet Modification and Balloon Valve Fracture
Key Summary
• In an 81-year-old man with valve-in-valve-in-valve TAVR and high coronary obstruction risk, a strategy combining leaflet modification, bailout techniques, and valve fracture enabled safe implantation and optimized postprocedural gradients.
• Multidisciplinary planning (interventional cardiology, cardiac surgery, imaging, anesthesia) is essential for complex TAVR, enabling shared decision-making, procedural precision, and safe real-time adaptation when strategies need to change.
• Advanced imaging, AI-based planning, and next-generation valve design will be key to improving durability, coronary access, and outcomes in repeat TAVR.
In this interview with JIC, Dr Apurva Patel discusses insights from the study, “Tailored Solutions for Structural Valve Degeneration: Valve-in-Valve-in-Valve TAVR With Coronary Leaflet Modification and Balloon Valve Fracture,” presented at the 2026 American College of Cardiology Scientific Sessions, highlighting evolving strategies for complex redo TAVR and lifetime valve management.

Hofstra School of Medicine
Northwell Health Lenox Hill Hospital
1. This case highlights a complex valve-in-valve-in-valve transcatheter aortic valve replacement (TAVR) with multiple adjunctive techniques—how did your prior experience with similar cases shape your procedural strategy and decision-making in this patient?
Great question—this case underscores why procedural planning with explicit contingencies is essential in complex valve-in-valve-in-valve interventions.
We evaluated an 81-year-old man with a prior surgical Mosaic valve (Medtronic) (2004) followed by valve-in-valve TAVR with a 23-mm Portico (Abbott) (2014) who presented with worsening dyspnea. Echocardiography showed severe structural valve degeneration with a mean gradient of 56 mm Hg, Vmax 4.68 m/s, and moderate aortic insufficiency. The central challenge, however, was not simply implanting a third valve—it was doing so safely and effectively. Preprocedural computed tomography (CT) demonstrated a left valve-to-coronary distance of only 3.3 mm, well below accepted safety thresholds, making coronary obstruction highly likely without leaflet modification.
Based on prior experience with similar anatomically high-risk cases, we proceeded with a primary strategy and a clearly defined backup plan. Plan A was leaflet modification using the ShortCut device (Pi-Cardia), which we felt was well-suited to this anatomy. However, we also planned for UNICORN as an alternative, recognizing that tortuous or severely angulated arches can make device delivery unpredictable.
That contingency proved critical. When we attempted to advance the ShortCut device, the severely angulated distal aortic arch (Figure 1C) prevented passage. We promptly pivoted to the UNICORN technique to achieve leaflet laceration, then performed valve fracture of the existing construct using a 24-mm True balloon (BD), followed by implantation of a 23-mm Sapien Ultra Resilia (Edwards Lifesciences).
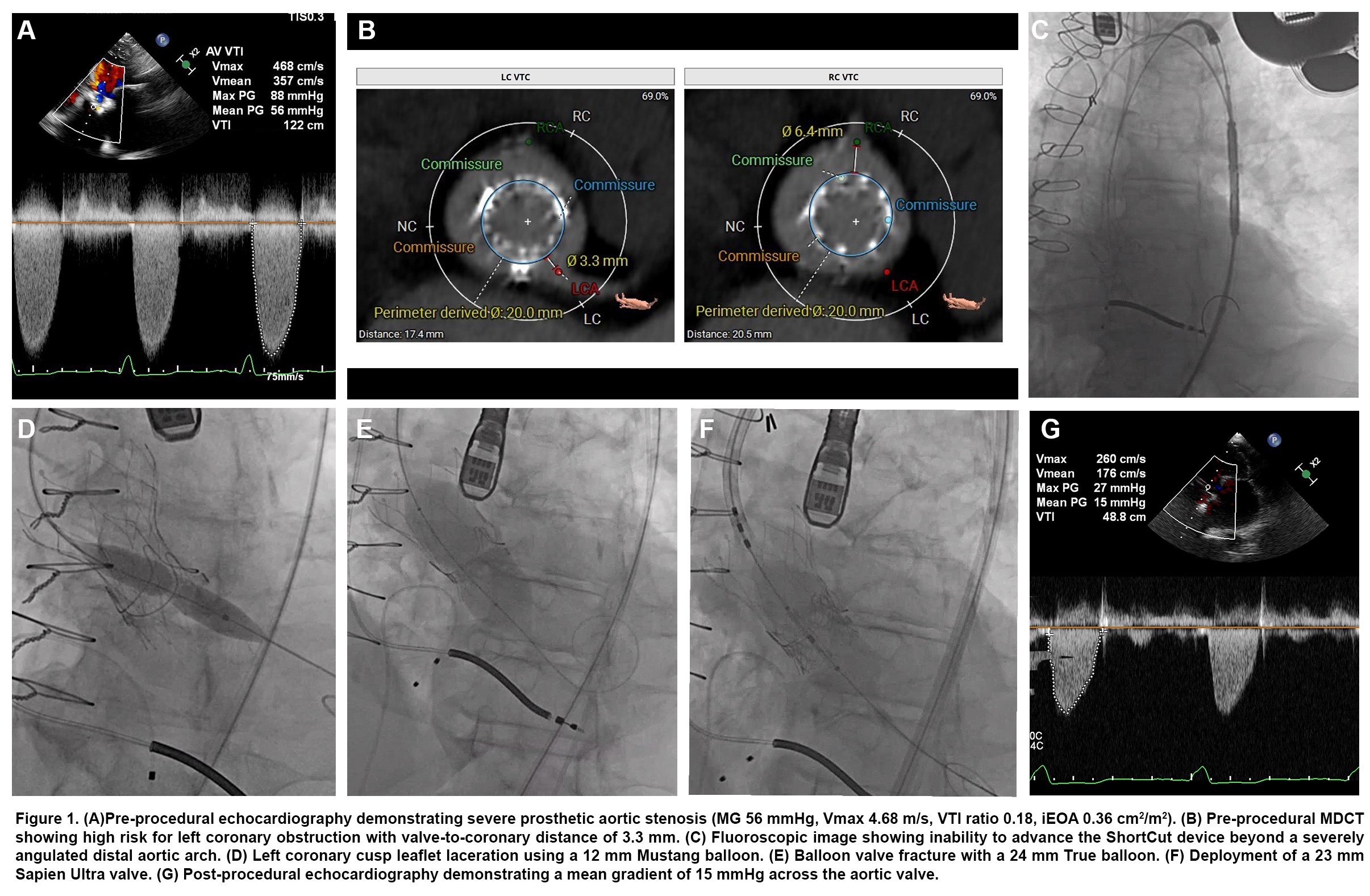
Final hemodynamics were excellent, with a mean gradient of 15 mm Hg—improving even on his post-2014 baseline of 20 mm Hg. The outcome reinforced the value of a deliberate sequence: leaflet modification for coronary protection, valve fracture for hemodynamic optimization, and then valve implantation—an approach refined through experience with these challenging redo cases.
2. Given this patient’s long clinical history, and that of others you have treated, how is your center approaching lifetime management planning for TAVR patients across both early and late stages of intervention?
This may be the most consequential question in structural heart disease today, because this patient’s course—surgical AVR (SAVR) in 2004, valve-in-valve TAVR in 2014, and now valve-in-valve-in-valve in 2025—reflects a 21-year trajectory that will become increasingly common.
At our center, we’ve fundamentally shifted to a lifetime-management mindset. We no longer view any valve procedure in isolation. Each decision—from the first implant to every subsequent reintervention—is made with the next step in mind. In that framework, SAVR and TAVR are not endpoints; they are tools within a longer strategy.
Practically, that approach includes several key elements:
First, imaging is the backbone. We maintain comprehensive longitudinal imaging for each patient, including serial echocardiography, CT datasets, and hemodynamic records. When this patient returned, we were able to compare the current CT with prior studies, define how the nested prostheses were oriented, precisely measure the valve-to-coronary distance (3.3 mm on the left), and evaluate the arch anatomy that ultimately drove an intraprocedural pivot. Without that infrastructure, planning is inherently incomplete. For first-time patients, we also incorporate artificial intelligence (AI)-based planning tools to simulate valve deployment and anticipate future reintervention scenarios.
Second, the Heart Team model is non-negotiable. This case was reviewed in a multidisciplinary conference including interventional cardiology, cardiac surgery, advanced imaging, and cardiac anesthesia. The conclusion that redo surgery was prohibitively high-risk, the strategy of leaflet modification plus valve fracture, and the contingency planning were all products of collaborative decision-making rather than a single-operator plan.
Third, we think differently about valve selection from the very beginning. When selecting a surgical valve or a first TAVR, we focus on factors that directly influence future options: true internal diameter, feasibility of valve fracture if expansion is needed later, commissural alignment, and the projected coronary relationship after an additional nested valve. These considerations are no longer theoretical—they directly determine what will be safe and feasible 10 to 15 years later.
Finally, we invest in patient education and longitudinal follow-up. Patients need to understand that bioprosthetic valves—surgical or transcatheter—have a finite lifespan. A core part of our role is to set expectations early, plan the long-term pathway with the patient, and ensure durable follow-up so reintervention can be timed before decompensation and performed under optimal conditions.
3. More broadly, as more patients outlive their initial bioprosthetic and transcatheter valves, how do you see the field evolving in terms of device design or procedural planning to better accommodate repeat interventions like valve-in-valve-in-valve TAVR?
This case highlights both the promise of contemporary transcatheter therapy and the limitations of applying first-generation design concepts to an era of repeat interventions. It also points to several clear priorities for where the field needs to go next.
First, valve platforms will need to be designed with repeat intervention as a primary use case. Today, we are frequently implanting valves that were engineered for a single lifetime implant and then asking them to accommodate a second—and increasingly a third—prosthesis. Future transcatheter heart valve designs should incorporate open-cell or coronary-friendly frame architecture to preserve coronary access, along with reliable commissural alignment features and visible markers to facilitate predictable “stacking” during redo procedures. In parallel, emerging TAVR designs—such as leaflets manufactured from a single piece of tissue to better mimic native valve function—may offer incremental hemodynamic improvements, which become especially important as effective orifice area is progressively constrained with each nested valve.
Second, leaflet durability remains a central frontier. If we can extend bioprosthetic durability from the current 10- to 15-year expectation to 20 years or longer, we could substantially reduce the number of patients who ever reach a third or fourth intervention. From a population standpoint, improving durability may be the single highest-impact innovation.
Third, computational planning is likely to become indispensable. With multiple nested prostheses interacting with native anatomy, the geometric complexity often exceeds what clinicians can reliably infer from conventional imaging alone. Patient-specific computational tools—finite element modeling, AI-enabled CT analysis for automated valve-to-coronary distance measurement and obstruction risk prediction, and even 3D-printed models for rehearsal—will increasingly guide strategy selection and procedural sequencing.
Finally, the evidence base must mature to match the clinical reality. We need longer follow-up in trials—not just 5 years, but 10 to 15 years—and dedicated valve-in-valve registries that systematically capture anatomy, device platforms, leaflet modification strategies, coronary access outcomes, and long-term durability. Individual cases are informative, but only large-scale longitudinal data will identify which approaches provide the safest and most durable lifetime pathway.
4. Based on your growing experience with these types of interventions, what key lessons or best practices would you share with other centers aiming to safely take on more complicated structural procedures, particularly in older, high-risk patients?
If I had to distill our experience into advice for other centers, I'd highlight 5 key principles.
First — and this is the most important lesson from this case — explicit planning and always have a contingency plan. In complex structural intervention, Plan A failing is not a complication; it's an anticipated possibility. Before this case, we had ShortCut as our primary leaflet modification strategy and UNICORN as our backup. The entire team was briefed on both approaches. When ShortCut couldn't be delivered, we pivoted seamlessly. If you walk into the lab with only one plan, you're setting yourself up for a crisis.
Second, invest heavily in imaging expertise. The pre-procedural CT in this case was not just helpful — it was essential. The 3.3 mm valve-to-coronary distance told us we must do leaflet modification. The arch anatomy warned us that device delivery could be challenging. Centers that want to take on these cases need dedicated structural CT readers who understand valve geometry, coronary relationships, and the specific measurements that drive decision-making. You cannot do complex structural work without world-class imaging.
Third, build complexity gradually. There’s a natural progression: first master standard valve-in-valve procedures, then incorporate balloon valve fracture to optimize expansion and gradients, then develop proficiency with leaflet modification—both electrosurgical and mechanical techniques (BASILICA, UNICORN, ShortCut)—and only after that begin combining these tools in the most complex redo scenarios. This stepwise pathway ensures each skill is reliable on its own before it is layered into higher-risk, multi-technique interventions.
Fourth, the heart team is not optional — it's essential. These cases require input from interventionalists, surgeons, imagers, and anesthesiologists. The decision to pursue a percutaneous approach, the risk stratification, the procedural planning, and the real-time intraprocedural decision-making all benefit from multidisciplinary collaboration. No single operator, regardless of skill, can optimize all of these dimensions alone.
And fifth, never accept a suboptimal hemodynamic result when you have tools to improve it. This patient had a baseline gradient of 20 after his prior valve-in-valve. Simply placing another valve without fracture might have produced severe prosthesis-patient mismatch — a gradient that could negate any clinical benefit. By combining balloon valve fracture with our valve deployment, we achieved a final gradient of 15 — better than his prior baseline. We owe it to these patients, who have been through multiple interventions and carry significant procedural risk, to ensure that when we do intervene, the result is optimized.
Ultimately, this case reinforces that lifetime management of structural valve degeneration is here, it's real, and it demands that we as a community continue to innovate, collaborate, and share our experiences — which is exactly why cases like this are so important to present and discuss.
Dr. Apurva Patel is a Structural Heart Interventional Cardiologist at Northwell Health Lenox Hill Hospital in New York City. His clinical and research expertise centers on the innovative treatment of complex structural heart conditions, including advanced transcatheter aortic valve implantation, mitral and tricuspid valve interventions. Dr. Patel is dedicated to improving patient outcomes through minimally invasive, catheter-based techniques, advanced imaging, and comprehensive lifetime management strategies for valvular heart disease. He is actively involved as an investigator in cutting-edge clinical trials for various structural heart procedures. A prolific author, Dr. Patel has contributed to numerous peer-reviewed publications and book chapters. He is a speaker at regional, national, and international conferences, where he shares his insights on topics including TAVR, TMVR, TTVR, advanced coronary imaging, and complex PCI. His significant contributions have been acknowledged with multiple honors and awards throughout his distinguished career. He is a Fellow of both the American College of Cardiology (FACC) and the Society of Coronary Angiography and Interventions (FSCAI).
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of the Journal of Invasive Cardiology or HMP Global, their employees, and affiliates.