Beyond the LRINEC Score: A Fatal Case of Vibrio vulnificus Necrotizing Fasciitis
Vibrio vulnificus necrotizing fasciitis is a rapidly progressive, often fatal soft tissue infection that demands immediate clinical recognition and decisive surgical management. This case underscores the limitations of relying on the LRINEC score in Vibrio-specific infections and highlights how early clinical findings must drive intervention. For podiatric surgeons, timely diagnosis and aggressive source control can mean the difference between limb salvage and loss of life.
Key Takeaways
1. Do not rely solely on the LRINEC score in suspected Vibrio infections. Vibrio vulnificus necrotizing fasciitis frequently presents with low initial LRINEC scores, demonstrating poor sensitivity and risking dangerous delays in surgical intervention.
2. Hemorrhagic bullae and rapid progression are red flags. Disproportionate pain, violaceous discoloration, and rapidly spreading edema—especially in patients with sepsis—should prompt immediate concern for necrotizing infection regardless of laboratory values.
3. Early, aggressive surgical management saves lives. Prompt radical debridement or proximal amputation, combined with targeted antimicrobial therapy, offers the best chance of survival in a disease where septic shock carries mortality rates exceeding 50%.
Vibrio vulnificus is a gram-negative bacilli bacterium found in warm salt water or shellfish. This organism has an association with fulminant necrotizing fasciitis (NF), especially in lower extremities exposed to seawater. Of note, patients with chronic liver disease are at an increased risk for sepsis due to this bacteria and even death.1,2 Clinically patients who have a Vibrio vulnificus infection present with bullae that often become hemorrhagic and progress to necrosis, septic shock, and multiorgan failure within hours. Pain is usually disproportionate in these patients who also exhibit signs of rapidly spreading erythema and edema.1,3
The Laboratory Risk Indicator of Necrotizing Fasciitis (LRINEC) score is a score originally derived from a cohort which included Vibrio cases, and has been widely used to assess the risk of NF infections.4 Although, literature shows that its diagnostic performance for Vibrio NF infection is inconsistent, limiting its use. Studies have shown a sensitivity of roughly 26–77% and specificity of 72–93%.5-7 A study by Hsiaio and colleagues in 2023 analyzed a cohort of 260 patients, 40 with Vibrio vulnificus NF infection and 80 with non-Vibrio NF infection.5 They found that patients who presented with Vibrio vulnificus infection that had a LRINEC cutoff more or equal to 6 had a sensitivity of 35% and specificity of 81%, concluding that LRINEC is not reliable to diagnose early Vibrio vulnificus NF infection but may have prognostic value for worse outcomes.5 Another study by Chao in 2012, analyzed 125 consecutive Vibrio vulnificus NF infection cases.6 Out of 125 patients, 72 were diagnosed with a NF infection. They found that hemorrhagic bullae and a low LRINEC threshold (LRINEC of more or equal to 2) were useful to flag NF infection in patients suspected of having a Vibrio vulnificus infection.6 More studies reveal that patients usually present with severe soft tissue infection despite the low LRINEC score, which oftentimes delays potentially life-saving surgery.8,9 When clinical suspicions arise, physicians should focus their diagnostic process on exposure history, clinical presentation (presence of bullae) and rapid progression of the infection.3
Initial Patient Presentation
This article presents the case of a 77-year-old female who presented to the Emergency Department on September 8, 2024 following an unwitnessed fall. At bedside, the patient appeared awake, but not alert or oriented to person, place, or time. The family at bedside denied any recent head strike or loss of consciousness. Of note, they stated that she placed her feet in hot water over the course of the past week, noting increased swelling and blistering to the left lower extremity for the past day. At baseline, the family stated she lives independently with her husband and is ambulatory with a walker.
The patient had a past medical history notable for type 2 diabetes mellitus, heart failure with preserved ejection fraction (HFpEF), severe pulmonary hypertension, moderate mixed valvular disease, coronary artery disease, atrial fibrillation, and was status post dual-chamber permanent pacemaker implantation and left atrial appendage occlusion with a Watchman device.
Emergency Department Course
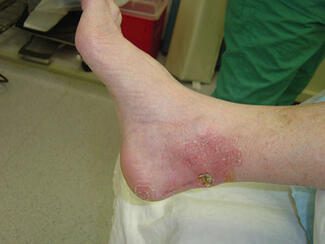
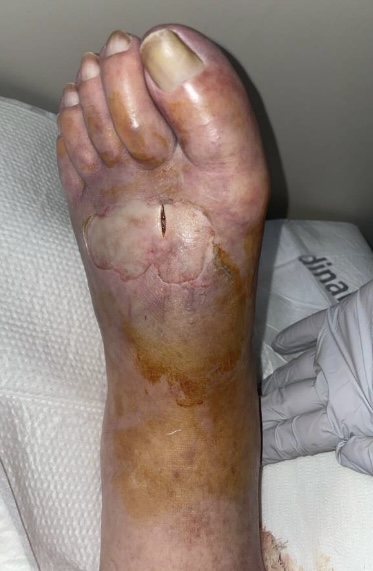
On arrival, the patient was febrile (temperature 39.2°C), tachycardic (heart rate up to 115 beats per minute), tachypneic (respiratory rate up to 39 breaths per minute), and hypoxemic (oxygen saturation 87% on 2 liters nasal cannula), requiring bilevel positive airway pressure ventilation. Electrocardiogram demonstrated atrial fibrillation with rapid ventricular response and QTc interval. Lower extremity clinical examination revealed violaceous hemorrhagic blistering and small pustules scattered over the medial calf and dorsal foot, with erythema and dusky cyanotic discoloration over the forefoot and medial calf (Figure 1). We did not note any findings of malodor, open wounds, active drainage, prominent fluctuance or soft tissue crepitus. Bilateral dorsalis pedis and posterior tibial pulses were nonpalpable, with delayed capillary refill time. The left limb was warm proximally, with the distal digits cool to touch. Gross sensation was diminished and +2 pitting edema was evident on the dorsum of the left foot.
Computed tomography of the left lower extremity demonstrated circumferential soft tissue edema and fascial thickening, without fluid collection, soft tissue emphysema, or osseous abnormalities. These findings were considered in the differential to represent volume overload, edema, or cellulitis. Chest radiographs showed pulmonary congestion and right predominant bilateral airspace opacities, suggestive of superimposed pneumonia.
Laboratory evaluation demonstrated leukocytosis (white blood cell count 26.02 x 109/L) and elevated inflammatory markers (erythrocyte sedimentation rate 47 mm/hr, C-reactive protein 10.24 mg/dL). The LRINEC score was 3, which included additional values of hemoglobin at 14.9 g/dL, sodium at 138 mmol/L, creatinine at 1.59 mg/dL, and glucose at 228 mg/dL. In addition, the remainder of the infectious work-up demonstrated a positive urinalysis, suggesting urinary tract infection.
Clinical Progression and Surgical Course
The patient began broad spectrum intravenous antibiotics, including piperacillin-tazobactam, vancomycin, and clindamycin, and vascular surgery and podiatry services consultations took place for evaluation of the progressing blistering lesions with concern for deep soft tissue infection. We performed bedside incision and drainage of the left medial calf and dorsal foot for source control, expressing approximately 10 cc serous clear drainage from the dorsal foot, and obtained wound cultures (Figure 2).
Blood and wound cultures subsequently grew Vibrio vulnificus. The patient then developed severe sepsis with septic shock, requiring medical intensive care unit (MICU) admission, vasopressor support, noninvasive positive pressure ventilation for acute hypoxemic respiratory failure, and central venous and arterial lines for hemodynamic monitoring. Antibiotic therapy adjustments took place per infectious disease recommendations to doxycycline, linezolid, and meropenem. A subsequently elevated LRINEC score of 8 raised concern for necrotizing infection.
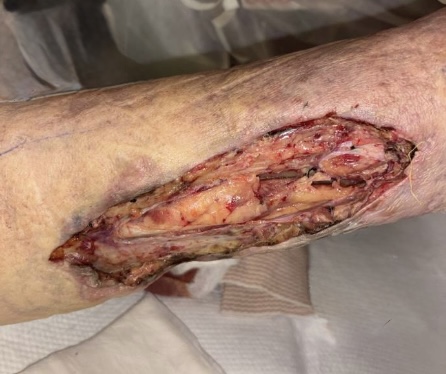
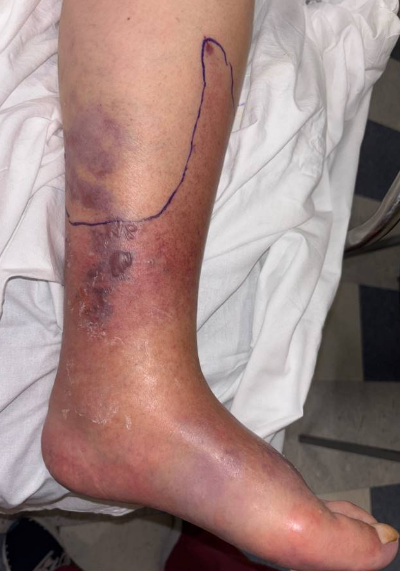
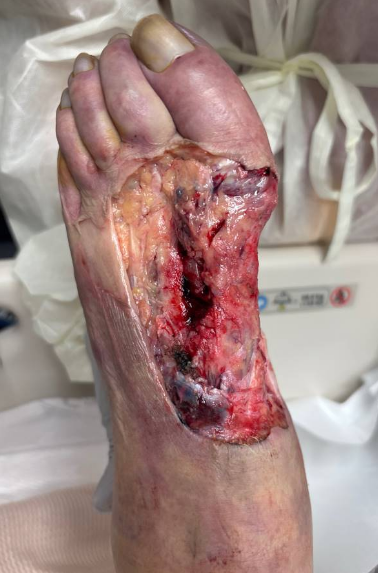
From a podiatric perspective, the foot was deemed nonsalvageable and a proximal amputation was recommended. After extensive treatment discussions with the patient’s family, the patient’s health care proxy declined amputation. The patient then underwent operative debridement with vascular and podiatry teams for aggressive source control of infection, while intubated on multiple vasopressor agents. Vascular surgery performed an elliptical medial distal calf incision (8 cm x 5 cm), with excision of skin, subcutaneous tissue, and fascia, revealing viable underlying muscle without purulence (Figure 3a). Podiatry then performed a dorsal elliptical incision over the first through fourth metatarsals, expressing 10 cc of purulence with nonviable, non-contractile intrinsic musculature. Extensive debridement resulted in a 15 x 10 cm defect (Figure 3b). Following meticulous hemostasis, the wounds were packed and dressed and the patient was returned to the MICU in critical condition, with consideration for further debridement or amputation pending the health care proxy’s consent.
Remainder of Hospitalization
The following day the patient developed a seizure-like episode characterized by right upward gaze and generalized shaking, treated with intravenous midazolam. Multi-organ dysfunction ensued, as the patient developed acute respiratory failure requiring mechanical ventilation, acute oliguric renal failure with severe metabolic and lactic acidosis necessitating continuous veno-venous hemodialysis (CVVH), and progressive circulatory collapse requiring rapid dose escalation of vasopressors.
Hemodialysis was eventually discontinued due to poor tolerance and unstable hemodynamics. Her course was further complicated by persistent atrial fibrillation with rapid ventricular response, requiring amiodarone infusion, and malignant ventricular arrhythmia in the setting of worsening acidosis and hyperkalemia. At this time, laboratory studies were consistent with persistent multi-organ failure with concern for disseminated intravascular coagulation and intravenous antibiotics were continuously adjusted per infectious disease recommendations.
Three episodes of ventricular tachycardia cardiac arrest ensued, requiring advanced cardiac life support and defibrillation. In between episodes, the patient received maximal medical management with potassium-lowering therapies, sodium bicarbonate, and continued antiarrhythmic infusions. Despite achieving return of spontaneous circulation, the patient’s poor baseline cardiac function, overwhelming bacteremia in the setting of necrotizing fasciitis, and multiple cardiac arrests, all indicated a very poor prognosis. The patient ultimately suffered an asystolic cardiac arrest, requiring cardiopulmonary resuscitation, which was eventually discontinued at the family’s request. The patient was pronounced deceased at 12:50 PM on September 13.
Discussion
This case highlights the serious and fatal nature of Vibrio vulnificus necrotizing fasciitis, especially in elderly patients with significant comorbidities. This patient’s course demonstrates how immunocompromising states, such as diabetes mellitus, as well as severe cardiac dysfunction, can precipitate rapid systemic collapse. The rapid progression from presentation to septic shock and eventual mortality in this 77-year-old female reinforces the importance of a high index of suspicion as well as early and aggressive surgical intervention, often requiring amputation over limb salvage.
Diagnostic Challenges and the LRINEC Score. An important and often overlooked teaching point is the limitation of the LRINEC score in diagnosing Vibrio infections. Upon initial presentation, this patient had a LRINEC score of 3, which stratifies as "low risk" for NF. However, and most importantly, she also exhibited classic clinical signs, including violaceous hemorrhagic blistering and rapid symptom progression. This discrepancy aligns with the literature cited by Hsiao and Chao, and their respective teams, which demonstrates that the LRINEC score has poor sensitivity (26–77%) for Vibrio-specific infections.4,5
Chao in 2012 also noted that patients with V. vulnificus often present with severe soft tissue infection despite low initial inflammatory markers.4 As experienced in this case, the patient’s LRINEC score only elevated to 8 after she was already in severe septic shock. This further underscores the importance of prioritizing clinical findings, specifically the presence of hemorrhagic bullae and rapid discoloration, over laboratory scoring systems. Physicians, therefore, should steer clear of relying on a low LRINEC score, which can lead to dangerous delays in surgical intervention.
Clinical Presentation and Course. Hemorrhagic bullae are a hallmark of V. vulnificus septicemia. Bross and colleagues describe how the infection causes vasculitis, leading to ischemia, bullae formation, and necrosis.3 In this case, the transition from localized soft tissue findings to systemic multi-organ dysfunction syndrome was extremely rapid. The patient’s history of soaking her feet in hot water serves as a reminder that while seawater exposure is the classic vector, thorough history taking is vital.
Surgical and Medical Management. The mortality rate from V. vulnificus sepsis is high, particularly when hypotension develops, with mortality rates exceeding 50% in cases of septic shock.1 "Time is tissue" is a critical concept in NF; however, in Vibrio cases, time is also life, further necessitating early and timely intervention.
Current guidelines recommend a synergistic approach of aggressive surgical debridement and targeted antimicrobial therapy. In this case, the patient was appropriately transitioned to the correct antimicrobial regimen. However, the necessity for prompt, definitive surgical source control was undermined by the clinical presentation. During the initial incision and drainage, the minimal purulence expressed led to an underestimation of the infectious severity. This outcome highlights a critical diagnostic pitfall: the failure to recall that in early NSTI, the core pathology resides within the deep fascial planes rather than manifesting as a superficial abscess. Consequently, this observation contributed to a delay in the requisite surgical intervention, which was further complicated by the family’s refusal of amputation.
The decision regarding amputation versus debridement is complex. While the family initially declined amputation, favoring debridement, literature suggests that early, aggressive radical resection is associated with a higher survival rate. The rapid progression of the disease, as seen in the increase in purulent drainage and intrinsic muscle necrosis in the operating room, illustrates the aggressive and fast proximal spread of this bacteria.
Conclusion
This fatality underscores that Vibrio vulnificus is a medical and surgical emergency that requires immediate recognition independent of risk stratification scores like LRINEC. For the podiatric surgeon, the presence of hemorrhagic bullae in a septic patient should trigger immediate broad-spectrum antibiotics and urgent definitive surgical intervention. In patients with significant comorbidities and rapidly deteriorating hemodynamics, early proximal amputation may offer the only chance of survival.
Drs. Llazar, Messiha, and Vishnubhatt are podiatric residents at New York Presbyterian Queens Hospital in New York City.
References
1. Haftel A. Vibrio vulnificus Infection. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2023.
2. Muldrew KL, Miller LG, Schmidhofer M, et al. Necrotizing fasciitis from Vibrio vulnificus: case report and literature review. J Clin Microbiol. 2007;45(9):2829-2832. doi:10.1128/JCM.00522-07
3. Bross MH, Soch K, Morales R, Mitchell RB. Vibrio vulnificus: diagnosis and treatment. Am Fam Physician. 2007;76(4):539-544.
4. Chao WN, Tsai SJ, Tsai CF, Su CH, Chan KS, Lee YT, et al. The Laboratory Risk Indicator for Necrotizing Fasciitis score for discernment of necrotizing fasciitis originated from Vibrio vulnificus infections. J Trauma Acute Care Surg. 2012;73(6):1576-1582. doi:10.1097/TA.0b013e318270d761
5. Hsiao CT, Weng HH, Yuan YD, Chen CT, Chen IC. Prospective validation of the Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) score for early diagnosis of necrotizing fasciitis. PLoS One. 2020;15(4):e0231873. doi:10.1371/journal.pone.0231873
6. Tarricone A, Tanini I, Pignatti G, et al. Diagnostic accuracy of the Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) score: a systematic review and meta-analysis. Surg Infect (Larchmt). 2022;23(6):459-469. doi:10.1089/sur.2021.196
7. Sato EH. The Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) score: review and clinical application. Acute Med Surg. 2020;7(1):e558. doi:10.1002/ams2.558
8. Chang CP, Hsiao CT, Weng TI, Chen IC, Lin CM, Liao CI, et al. Unreliable diagnostic accuracy of Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) score but good outcome predictor in necrotizing fasciitis due to Vibrio vulnificus: a retrospective and matched-pair study. Medicine (Baltimore). 2023;102(27):e34073. doi:10.1097/MD.0000000000034073
9. Liao CI, Lee YT, Su YC, et al. Validation of the Laboratory Risk Indicator for Necrotizing Fasciitis score in a general population. J Microbiol Immunol Infect. 2012;45(6):395-399. doi:10.1016/j.jmii.2012.03.008
© 2026 HMP Global. All Rights Reserved.
Any views and opinions expressed are those of the author(s) and/or participants and do not necessarily reflect the views, policy, or position of Podiatry Today or HMP Global, their employees, and affiliates.